The semester is over, final grades are submitted, and all but the graduating students have left campus. At this point, students of Ichthyology may be
more frustrated and less fascinated by the enormous diversity of fishes. In lab we introduced students to over 74 of
the 515 families of fishes in the world.
From the monospecific Amiidae
(bowfin) to the species-rich Cyprinidae (minnows and carps), each group of
fishes presents different challenges for identification. Some,
such as the gars are so unique that you will never misidentify one, whereas
others, in particular the sculpins, are easily confused even by the experienced
fish specialists. The week before the
final lab practical we visited and sampled two tributaries of the New River
drainage. Over 89 fish taxa are known in the upper New
River drainage and 48 are native and 8 are endemic. When sampling a single reach we are, in
fact, zooming in from the zoogeographic pool of likely fishes to the specific
assemblage that is present at the site.
Here local factors, such as elevation, channel slope, temperature, and
physical habitat will influence the fish species we encounter. Small
high-elevation mountain drainages may have only a few species, such as brook
trout, blacknose dace, and mottled sculpin, while the larger downstream reaches
may support 12 to 15 species. In diagnosing fish species, I also preach
the value of zooming in.
 |
| Photo of Blacknose Dace Rhinichthys atratulus, Sinking Creek near Newport, Virginia. Photo: D. J. Orth |
For weeks the students were learning characteristics and annotated their specimens based on examination of specimens preserved in ethanol. Often the name was in the jar - no challenge there. As I watched the students faced with an unknown live and
squirming specimen, I encouraged them to “Tell me what you see. What do you
think it is?” Students have been
learning to start with the bigger categories and drill down to likely species
by zooming in on diagnostic characteristics. For example, the minnows can be bewildering
until the student learns to zoom in on characteristics of the mouth, or scale
size and pattern. Most minnows have a
small terminal mouth, so any variation from this is important. A ventral mouth may mean a Rhinichthys dace, Kanawha minnow,
cutlips minnow or central stoneroller.
Zooming in on the characteristics of the mouth will quickly reveal the
diagnostic character of the species. For example, the cutlips and tonguetied minnows are distinguished for all other minnows by its three-lobed lower jaw with middle lobe sticking out like a tongue (photo on left). This adaptation allows it to capture miniscule gastrods and insect larvae, which it scrapes from rocks, and also assists the male cutlips minnow who builds a nest of small pebbles; click here to view a video. The lower jaw of the Central Stoneroller has a hard cartilaginous ridge (photo on right) which it uses to scrape ooze from the stream bottom. It gets its name by the habit of the male excavating a nest by moving gravel with its nose.
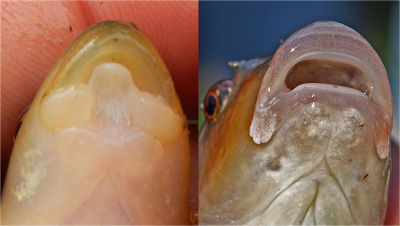 |
| Close-up photos of the ventral view of the mouth of Cutlips Minnow Exoglossum maxillingua (Photo by Dylan Hann) and Central Stoneroller Campostoma anomalum (Photo by D. J. Orth.) |
It is spring time and the males of many species are showing
the special secondary sexual characteristics.
The Nocomis chubs and Campostoma stonerollers already have
tubercles on the heads, and white suckers and hog suckers have tubercles on
anal and caudal fins. The male
fantail darter Etheostoma flabellare develops little egg mimics at the tips of his
spiny dorsal fin. The fantail darter selects a large flat
cobblestone in riffles for a nest site and the female lays eggs on underside of
the cobble while the male fertilizes and guards the developing embryos. The egg mimics are a bit of visual trickery as the females searching for a suitable mate and nest site see there are already eggs in the nest. In fact, in an elegant experiment, Knapp and Sargent (1989) demonstrated that females preferred males with eggs over males without eggs, and furthermore that females preferred males with egg-mimics over males without egg-mimics. Therefore, female choice played a major role in the evolution of the egg-mimics in fantail darters.
The week after our field trips, a spring
storm dumped heavy rains and these easily-waded streams now overflowed their
banks. The large flat cobblestones
selected as nest sites by the fantail darter are highly resistant to being
entrained and mobilized in these common spring floods. The cavity under the cobblestone is also inaccessible to potential egg predators. I am certain the less-selective male
fantail darters soon learn the hard way the value of finding a nest site below a big, stable cobblestone.
 |
| Fantail darter Etheostoma flabellare from Sinking Creek near Newport, Virginia. Note the egg mimics at tips of spines of first dorsal fin in this male specimen. Photo by D. J. Orth |
The sculpins (Cottidae) are a group of bottom-dwelling fishes that are widely distributed in temperate regions of North America and Eurasia. They exhibit extreme variation in morphology and species are difficult to characterize and identify. While the group is not as species-rich as either the
darters or minnows, their identification may stymie the novice
Ichthyologist until s/he learns to zoom in on the characteristics. Streams we sampled had both the Mottled
Sculpin Cottus bairdi and the Kanawha
Sculpin Cottus kanawhae (formerly
considered a subspecies of banded sculpin Cottus
carolinae carolinae Robins 2005).
Here I include close-up photos to show the pigmentation patterns of the dorsal fins and chins.
 |
| Dorsal fins of the Kanawha Sculpin (top) and the Mottled Sculpin (bottom). Photo by D. J. Orth |
Sculpins are so well camouflaged to match the stream bottoms that anglers are seldom aware that the streams they fish are loaded with sculpins. Some fly tiers have learned to mimic the color and pattern of the sculpin with streamer patterns. In fact the world-wide favorite Muddler Minnow was created to mimic a sculpin. These are effective for a variety of game fishes as long as they are fished right on the bottom and fished slowly, with occasional twitches to mimic escape action.
 |
| Chins of the Mottled Sculpin (left) and the Kanawha Sculpin (right). Photos by D. J. Orth |
The family Cyprinidae (minnows and carps) is one of the largest families of fish in the world (over 2,400 species in the last authoritative count) and these are often the most abundant fishes in freshwater streams. The diversity of the Cyprinidae is truly impressive on a global scale and when we zoom in on the Virginia waters we have 67 species. All the minnows have one dorsal fin, pelvic fin in abdominal position, pectoral fin low on the body, and lack an adipose fin. Beyond that one needs to zoom in the lower jaw, tiny barbels, pigment patterns, scale size and pattern, and even fin ray counts. The first fish we captured in Toms Creek was a very small minnow, a rosefin shiner Lythrurus ardens. I didn't think the students would be able to identify it, but when they zoomed in to observe the diagnostic characteristics, they nailed it.
 |
| Telescope Shiner Notropis telescopus from Sinking Creek near Newport. Photo by D. J. Orth |
 |
| White Shiner Luxilus albeolus from Sinking Creek near Newport. Photo by Jessica Dodds. |
 |
| Close up photos of the dorsal scales of the Telescope Shiner (top) and lateral line scales of the White Shiner (bottom). Photos by D. J. Orth. |
Telescope shiner and white shiner are depicted in these photos --
when I first learned Ichthyology these two were in the same genus Notropis, a lot of things were easier back then. But when we zoom in, to the scale patterns we see important differences. Telescope shiner had dark scale margins in the nape and dorsolateral scales and the scales are larger and irregularly shaped, leaving a distinct zig-zag line pattern. The White Shiner is in the genus of high-scale shiners (Luxilus) so named for the exposed scales in lateral line that are twice as high as the exposed width -- something you only notice when you zoom in.
We encountered two species of Rhinichthys, the daces. The genus name Rhinichthys translates to "snout fish" in reference to the prominent snout. Here zooming in requires the observer to be more precise about
the pattern or proportions.
The blacknose dace has a blacknose, the longnose dace has a longnose,
yet the correct identification requires us to be more precise about “how long
is the long nose?” We must zoom in to
interpret. The two species co-occur in Ridge-and-Valley and Blue Ridge streams and are often easy to confuse.
 |
| Photos of head (lateral view) and ventral view of mouth of Longnose Dace Rhinichthys atratrulus. Photos by D. J. Orth. |
There is much value in zooming in and ever since my close-up
vision was compromised by middle-age, I have had to rely on a magnifying lens to
assist me when zooming in. There is plenty to do while sampling for fish in our local streams or sorting through a net full of fish. Zooming seems like one of the last things to do after obtaining a sample, but it is an essential part of the fascinating study of the fishes. To my Proud Ichthyology Students of 2013, go forth and catch many fishes, use your newfound knowledge to earn an honest living, share your enthusiasm for fish and their habitats with family and friends, never be satisfied with the status quo, and know when you need to zoom in.
References
Knapp, R. A., and R. C. Sargent. 1989. Egg-mimicry as a mating strategy in the fantail darter, Etheostoma flabellare: females prefer males with eggs. Behavioral Ecology and Sociobiology 25:321-326.
Knapp, R. A., and R. C. Sargent. 1989. Egg-mimicry as a mating strategy in the fantail darter, Etheostoma flabellare: females prefer males with eggs. Behavioral Ecology and Sociobiology 25:321-326.
Robins, C. R. 2005. Cottus
kanawhae, a new cottid fish from the New River systems of Virginia and West
Virginia. Zootaxa 987:1-6.









